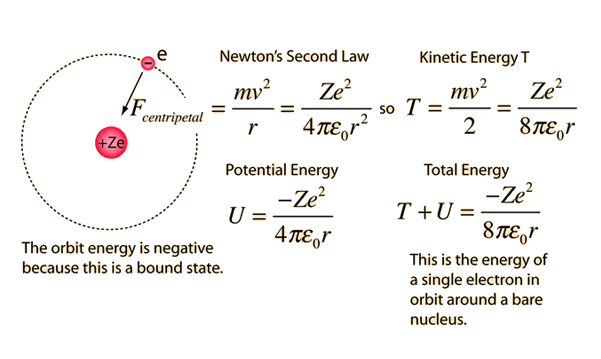
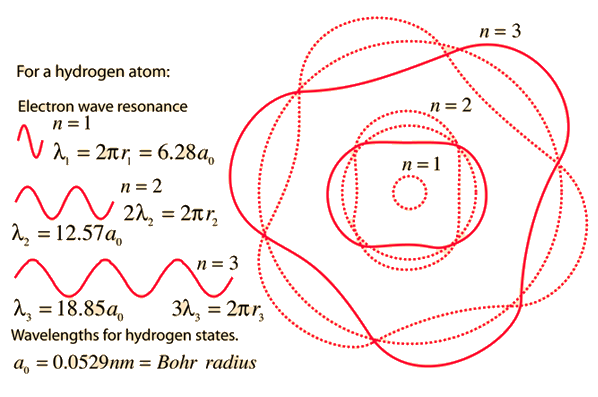
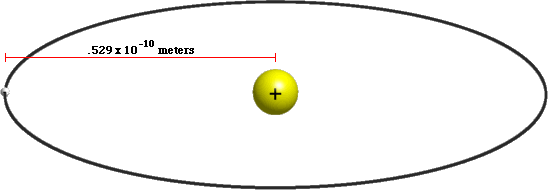
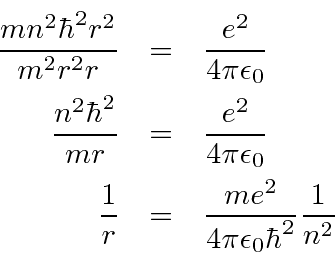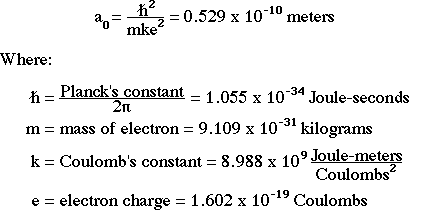PPT - 1. The first Bohr's radius (for electron in hydrogen atom in the ground state): PowerPoint Presentation - ID:6524739

Question Video: Understanding the Effects of Electron Mass on Orbital Radius Using the Bohr Model | Nagwa

MathType - The most probable distance between the nucleus and the electron in a hydrogen atom in its ground state is given by the Bohr Radius. This physical constant is named after
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community
What is the most possible radius (in PM) for an electron in the first orbit of a hydrogen atom? - Quora