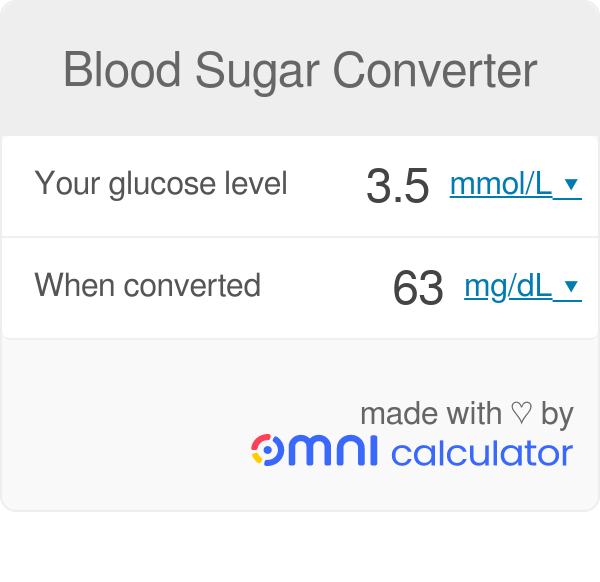
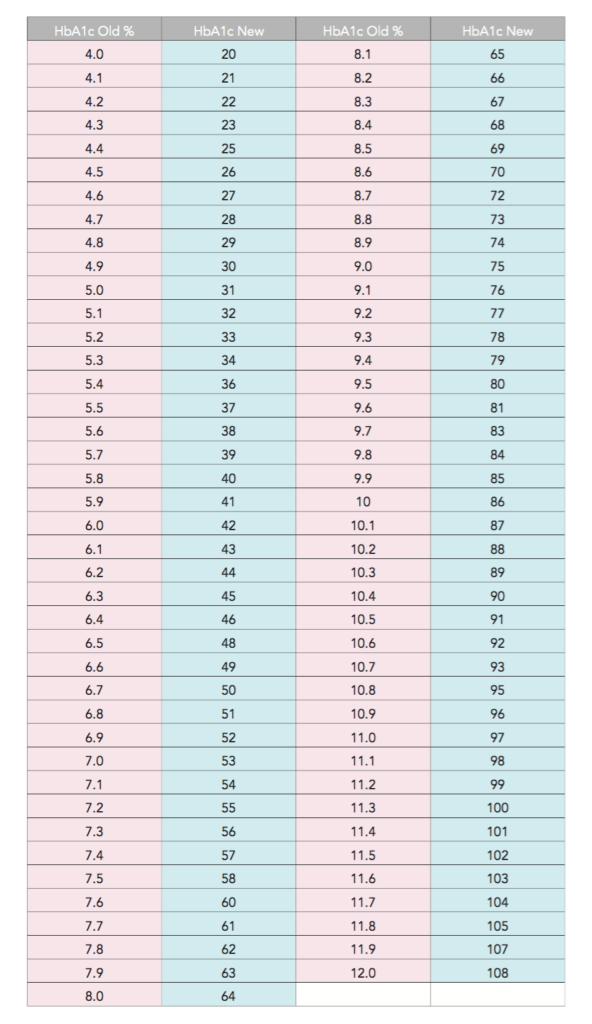
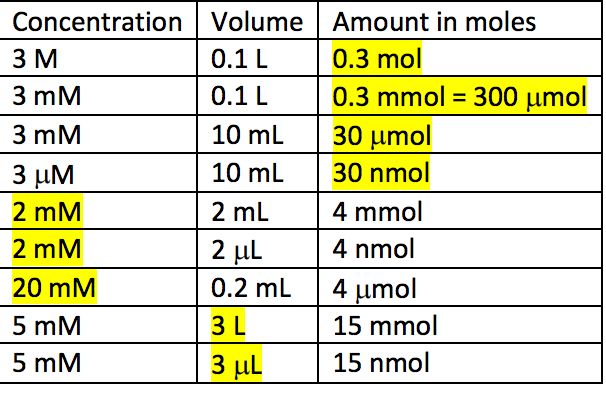
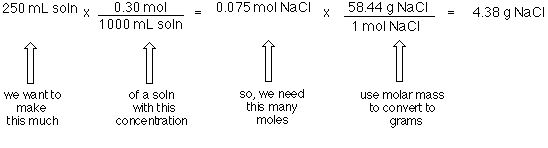Stomatal conductance are often expressed in units of mol/(m**2 s). How do we convert into units of m/s for meteorological applications?

Calculations Involving Concentrations The stoichiometry calculations done in Chapter 8 involved pure substances and samples whose mass could be determined by weighing. In Chapter 9, the reactant was often a gas. In these cases the mass of the sample ...

8/1/20151 Chapter 3 Stoichiometry 3.1 Atomic Masses 3.2 The Mole 3.3 Molar Mass 3.4 Percent Composition of Compounds 3.5 Determining the Formula of a. - ppt download





.png)